mercury & photography
Written by Jesse Andrewartha
DISCLAIMER: Mercury and mercury compounds are extremely toxic and include some of the most highly regulated reagents in the world. Poisoning can result in serious long term injury and death. Always use in accordance with local environmental ordinances and hazmat/whimis regulations with appropriate personal protection. While I have detailed protocols and handling in this article, it is incumbent on any person seeking to use mercury and its compounds for any reason to conduct full research and understand all local laws relating to their possession, use and disposal. The author is not responsible for any accident, contamination, injury or death resulting from misuse or possession of mercury or mercury compounds.
INTRODUCTION
Mercury and its compounds have been a subject of both wonder and utility throughout history. Over the centuries, they have been used in alchemy, for industrial processes, to mine gold, to end life by suicide or murder and as desperate medicine for treating illness. The silvery metal gave rise to history’s first mandates toward occupational health and safety for those who mined it, and although its toxicity has reduced its use in modern applications, mercury's contribution to significant scientific discoveries is undeniable. It was pivotal in identifying 22 elements, including oxygen, nitrogen, magnesium, aluminum, and helium. However, its role in the advent of photography perhaps remains one of its most profound impacts.
There is considerable controversy around the use of mercury in the darkroom. Discussions on modern photography forums ignite passionate debate and furious warnings, and not without reason: there is significant potential for damage and injury, both to the environment and to people, with the use of mercury and its compounds. However, the element endures as one of the most curious and unique additions to darkroom history and lore, and its use throughout the last 188 years of imaging is a remarkable story of discovery and danger which deserves to be told.
PHYSICAL CHARACTERISTICS
Mercury, the only metal that’s liquid at room temperature
Mercury, known by the symbol Hg, derived from its Latin name hydrargyrum, lies at atomic number 80 in the periodic table. It is a silver-white, heavy, mobile, liquid metal, the only metal that remains liquid at room temperature. It is 13.5 times denser than water, boils at 356.7°C, freezes at -38.9°C and in that state may be cut with a knife. Mercury amalgamates with several metals, including copper, silver, gold and aluminium.
In nature, it is a rare metal, with an average crustal abundance by mass of only 0.08 parts per million (ppm), generally found in combination with sulfur, forming the stable compound, cinnabar, though there are at least six common mercury-bearing minerals. The main source of mercury are mines located Spain, China, Kyrgyzstan and Algeria. The mines of Almadén, Spain, are some of the oldest mines of any kind in the world; approximately 250,000 metric tons of mercury were produced there in the past 2,000 years. It is not surprising that the unique metal would capture the imagination of the early photographic practitioners, who were often novice scientists and artists seeking to improve the speed and quality of their images.
EARLY PROCESSES
DAGUERREOTYPES
A daguerreotype of Louis Daguerre
The discovery of mercury development in daguerreotypy was actually an accident. The apocryphal story is that of Louis Daguerre at work in his laboratory in the 1830s. Daguerre had been collaborating with Nicephore Niepce, to improve the practicality of the heliotype, a notoriously slow process that required 8-12 hours to expose an image (See Image 1). Daguerre had eliminated the bitumen that was originally used to provide the shadow tones in the image, and had started experiments fuming silver plates with iodine to create a darkened surface. Frustrated one day by progress, he put several, seemingly blank, fumed and exposed plates in a cupboard, and when he returned the next day, he was stunned to find images upon the plates. A process of elimination over several days revealed beads of mercury as the cause for the appearance of the images.
When a Daguerreotype plate, sensitized with iodine & bromine and exposed to light, is subjected to mercury vapour, the mercury amalgamates with the light-sensitive silver halide surface of the plate, forming image particles that diffuse light, appearing white. These particles accumulate in proportion to the exposure of light and form a positive image on its surface (See Image 3). This discovery that a latent image, that is, a non-visible change to the sensitive surface by exposure to light, could be utilized and developed to create a full photographic image, revolutionized photography. It enabled the photographer to reduce exposure time to just seconds, the key that would become central to the daguerreotype’s commercial success.
The world’s first permanent photograph, a heliotype by Nicephore Niepce, formed by bitumen on a sheet of silver coated copper.
Mercury image particles on the surface of a daguerreotype plate under scanning electron microscope.
In the foundational book, “The Daguerreotype” by Barger and White, they describe image formation by mercury “as a vapor-phase mineralizer collecting both latent image (photolytic) silver and random photolytic silver and causing the metastable photolytic silver to recrystallize into stable silver image particles. Mercury plays the role of a solvent for crystal growth; it is not a chemical reactant in this process”.
The Daguerreotype would be the primary method of photography for over 20 years, finally succumbing to the ambrotype and tintype in the mid-1860s. But mercury’s role in photography was only just beginning.
Reverend Levi L. Hill.
THE HILLOTYPE
The Hillotype is an enigma in the history of photography. Invented by Reverend Levi L. Hill, it was the first colour photographic process, emerging five decades before the widely recognized advent of colour photography at the turn of the twentieth century. The Hillotype's history is fraught with controversy and allegations of forgery. For nearly 170 years, debates have persisted about whether Hill genuinely produced colour images. But the truth remains, the Hillotype is real, and in his discovery, Reverend Hill achieved something remarkable: he developed a colour photographic process that was decades more advanced than all of civilization's knowledge, allowing for the creation of permanent, natural color photographs before the US Civil War.The Hillotype utilized mercury for the first phase of plate preparation, as the white base onto which the sensitive surface would be formed. Starting with a daguerreotype plate, the photographer would prepare the plate by photographing an even, white surface. Developing the plate over heated mercury vapour, the resultant image formed white frosting of microscopic mercury particles; an even, white daguerreotype. Once fixed in the usual manner, this surface was immersed in a green, intensely acidic sensitizer mixed from a dazzling combination of hydrofluoric, hydrochloric and sulphuric acids with various metal salts. The sensitized surface would then be exposed to light in-camera. Much like the fledgling Daguerreotype process of the late 1830s, the Hillotype was incredibly slow, requiring exposures of at least 30 minutes in full sunlight. The latent image was developed using phosphuretted or sulphuretted hydrogen, with the final plate an amalgam of mercury, silver and the chemicals which make up the coloured specks of the image tone.
From Hill’s accounts of his process, he was evidently working largely by instinct when he invented the process and produced his first coloured images by accident in 1850. Having failed to fully account for the process, had no idea how he had photographed in colour. He spent the following three years immersed in his darkroom day and night, trying to recreate his formula. As he refined the Hillotype, Hill steadfastly refused to share his progress or his process, but tantalized the public by publishing press releases and sharing results privately with famous daguerreotypists of the time, such as Samuel Morse and Jeremiah Gurney. Such was the excitement for the potential new colour process, that the New York Daguerreian industry ground to a near halt, as clients stopped spending money on Daguerreotypes with the belief they could wait for the imminent release of the full-colour Hillotype. This prompted members of the New York State Daguerreian Association (NYSDA) to visit his house to demand answers; this devolved into an armed confrontation. The visitors threatened to destroy his lab and Hill drove off the officials with a handgun.
Joseph W. Boudreau’s modern Hillotype, created using the instructions from Hill’s “Treatise On Heliochromy”. The colours are true to the objects they depict, and correspond correctly on a Macbeth MS 1500 Spectrophotometer.
Hill penned a memoir on his experiences and processes in the incredible volume, “A Treatise on Heliochromy”. However, the book included Hill’s recounting of his experience with the officials from the NYSDA and he was sued for defamation. The courts agreed with the plaintiffs and the judge ordered all copies destroyed. It did not help that Hill had also hand-coloured parts of some of his Hillotypes, and his enemies seized upon those examples as evidence that he had not invented an automatic process, but rather was trying to pass off hand-coloured images as a true colour process. With his legacy shattered, the Hillotype was relegated as a footnote in history and its inventor discredited. Reverend Levi L. Hill died in 1865.
By incredible luck, several copies of “Treatise” survived and the treatise was reprinted in the 1960s. Then, in 1981, Professor Joseph Boudreau repeated Hill’s experiments following the instructions as provided in “Treatise”, successfully producing plates. Boudreau’s remarkable report on the experiments, "Color Daguerreotypes: Hillotypes Recreated", was published in 1984 in the anthology, “Pioneers of Photography: Their Achievements in Science and Technology”. In it, he detailed the incredibly dangerous process in a modern laboratory and provides several reproductions of his Hillotypes, which prove that Hill’s process does, in fact, produce full colour images. Several dozen of Hill’s original Hillotypes also survived, and are kept at the Smithsonian Institution.
LIPPMAN PHOTOGRAPHY
A Lippman photograph, 1899, by Dr. Neuhauss, a leading practitioner of the process at the turn of the 19th Century.
Lippman Photography is one of the first color processes. Invented in 1891 by Gabriel Lippman, this process doesn’t use any color dyes or color transfer. Instead, Lippman photography relies on the interference of light to create colour. The process relies on two distinct features: the use of an extremely fine grain, black and white emulsion and a special, modified film holder using mercury. The photographic plate is placed in the holder with the fine grain emulsion facing away from the lens and in optical contact with the mercury. Mercury was essential to creating this perfect mirror reflection required to bounce light through the emulsion within the film holder. When light passes through the emulsion and reflects of the mercury surface, the reflecting light waves interfere with the incident waves, forming a standing wave pattern within the volume of the emulsion.
Image formation in the Lippman plate. Mercury reflects light back through the emulsion, forming standing waves that are stored in the emulsion as density. (Source: Pénichon, Sylvie (2013): Twentieth Century Colour Photographs. The Complete Guide to Processes, Identification & Preservation. London, Los Angeles, Thames & Hudson, p. 235.)
The distance between these standing waves is a function of the colour, with reds, for instance, creating a greater distance than for blues, and these standing waves are stored in the emulsion as density, which is formed when the plate is processed. When viewed under white light in the correct conditions, the frequency of these densities reflect the colour of light that created them, resulting in a true colour image.
Lippmann plates never were used commercially, owing to their incredibly slow speeds and complexity in preparation, image capture and viewing. They have remained a curiosity, but a handful of photographers, such as contemporary Lippman plate photographer, Nick Brandreth, keep the process alive.
KELAINOTYPES
The kelainotype was the only pure mercury based imaging process ever created, invented in 1842 by Sir John Herschel. The kelainotype combined ammonium ferric tartrate and mercuric chloride, which reduced in the presence of UV light to create an image consisting of pure mercury metal. The image was reportedly incredibly beautiful and Herschel himself wrote of the kelainotype: "[the Kelainotype]… affords pictures of such force and depth of colour, such velvety richness of material, and such perfection of detail and preservation of the relative intensities of the light, as infinitely to surpass any photographic production I have yet seen, and which indeed it seems impossible to go beyond."
Unfortunately, due to the volatility of mercury metal, the image proves highly impermanent and to his chagrin, all of Herschel’s kelainotypes faded within a few days, provoking his frustrated comment: "Most unfortunately, they cannot be preserved." As expected, no specimens of a kelainotype exists today.
CYANOTYPES
A cyanotype of algae by 19th century botanist Anna Atkins
Cyanotypes were also invented in 1842 by Sir William Herschel. Combining potassium ferricyanide and ammonium ferric citrate, which reduce in the presence of UV light to prussian blue, or ferric ferrocyanide. In contrast to the kelainotype, the cyanotype was permanent. But mercury would still have a part to play:
In cyanotypes, mercuric chloride was experimented with by Herschel, who incorporated it into the sensitizer for a positive process. It is believed that it was successful because the process created prussian white, and mercury forms extremely stable complexes with cyanide. Therefore it bound the prussian white and prevented it reverting to prussian blue, an essential part of ensuring the longevity of a positive process cyanotype. Herchel’s efforts were not without reward: two wonderful specimens of his positive cyanotype process from 1842 survive and can be viewed in the collection of the National Media Museum at Bradford, UK.
KALLITYPES
Kallitypes is one of a number of printing processes in the iron-silver family, patented in 1889 by W. W. J. Nicol. These processes include methods such vandyke brown and argyrotype. In the Kallitype, ferric oxalate is combined with silver nitrate, which reduces in the presence of UV light to silver metal, but in each of these group of processes, the silver image is sensitive to the action of mercuric chloride, resulting in a tone that ranges from brown to olive. Unfortunately the use of this toner has one important negative effect: the image is more likely to stain and the print will be much more difficult to clear.
PLATINUM & PALLADIUM PRINTING
10080 counts per minute, Happy Jack Mine, White County Distric, Utah. Palladium print by Jesse Andrewartha, 2019
The platinotype process was invented by William Willis, who patented the process in 1873. The platinotype utilized platinum chloroplatinate and ferric oxalate, which reduced in the presence of UV light to form an image of pure platinum metal. Platinum images are generally neutral or even cool toned, and many photographers of the era weren't fans of their tonality. To address this, several companies started making sepia platinotype papers, giving the photos a much warmer, brownish tint. This was achieved by including mercuric chloride in the light-sensitive platinum coating on the surface of the paper. You could also get a similar effect by adding mercuric chloride to the developer. Mercuric chloride worked by slowing down the development of platinum particles in the image. The smaller size of the image particles affected how image absorbed and reflected light, making the final image look browner. The use of palladium as the imaging metal, or heating the potassium oxalate developer, were discovered to also contribute to a warmer tone, and eventually gained preference over the use of toxic mercuric chloride.
CONTEMPORARY PROCESSES
A NOTE ON TOXICITY
Mercury can cause both chronic and acute poisoning. Elemental mercury metal itself presents little danger; it is absorbed poorly through the skin or the gastrointestinal tract. In fact, mercury metal used to be used to clear intestinal blockages, owing to the weight of the metal! Once refined as elemental mercury, the liquid metal is volatile and forms a vapour at room temperature. It is this vapour that is the primary hazard of elemental mercury; once inhaled, the mercury easily passes into the body through the lungs, where it affects the brain and organs. An open container of mercury allowed to evaporate at 20°C in a closed room, until the air reaches equilibrium, will result in an environment that contains more than 100 times the current permissible exposure levels of mercury in a workplace. Daguerreotypists frequently fell ill to the effects of heated mercury vapour. It was known that practitioners could amalgamate a copper penny simply by rubbing it between their fingers, such was the amount of mercury in their sweat. Reverend Hill warned of mercury in “Treatise”: “Ventilate your mercury - its fume is loaded with rheumatism, sciatica, lumbago, tooth-ache, neuralgia and decreptitude”.
In contemporary photography, it is generally the water-soluble, inorganic mercury salts, mercuric(II) iodide or more commonly, mercuric(II) chloride, that are used. Of course, it bears repeating that soluble mercury compounds are extremely hazardous, both acutely and as a cumulative poison: mercuric chloride can be lethal at a dose of just 5 milligrams per kilogram of body weight, or about 400 milligrams for an average adult. Its toxicity is due not just to its mercury content but also to its corrosive properties, which can cause serious internal damage, including ulcers to the stomach, mouth, and throat, and corrosive damage to the intestines. Mercuric chloride also tends to accumulate in the kidneys, causing severe corrosive damage which can lead to acute kidney failure. However, mercuric chloride, like all inorganic mercury salts, does not cross the blood–brain barrier as readily as organic mercury, although it is known to be a cumulative poison.
Common side effects of acute mercuric chloride poisoning include burning sensations in the mouth and throat, stomach pain, abdominal discomfort, lethargy, vomiting of blood, corrosive bronchitis, severe irritation to the gastrointestinal tract, and kidney failure. Chronic exposure can lead to symptoms more common with mercury poisoning, such as insomnia, delayed reflexes, excessive salivation, bleeding gums, fatigue, tremors, and dental problems. Acute exposure to large amounts of mercuric chloride can cause death in as little as 24 hours, usually due to acute kidney failure or damage to the gastrointestinal tract. In other cases, victims of acute exposure have taken up to two weeks to die. Needless to say, it is a compound that demands the upmost respect.
SAFETY PROTOCOLS
If you are going to work with highly toxic mercury/mercury compounds in your darkroom, there are requirements for your working area to ensure your safety and for those around you:
1. FACILITY REQUIREMENTS
Fume hood tested at no less than 120 lfm average face velocity.
An eye wash station, placed no less than 10 seconds from the work area, with unobstructed access.
A shower for use in emergencies no less than 10 seconds from the work area.
Floors must have vinyl coverings, either tile or sheet.
A mercury spill kit must be kept in the darkroom with its location prominently displayed with a sign.
Always read the MSDS for all the chemicals involved in your process before purchasing them. Mercury compounds are only available for purchase by registered businesses with commercial addresses, and may require special equipment for handling/storage that you will need to acquire prior to their arrival. At all times practice strict chemical hygiene.
2. HYGIENE REQUIREMENTS
Do not eat, drink, smoke, or apply cosmetics in the darkroom.
Do not store food in darkroom refrigerators.
Wash hands and face after handling mercury and before breaks or finishing in your darkroom .
Avoid skin and eye contact — do not touch your face or rub your eyes in the darkroom.
Dress appropriately to work in the darkroom — long pants, closed shoes (no open toes, no canvas). Use appropriate Personal Protective Equipment (PPE)!
3. PPE REQUIREMENTS:
Safety glasses with side shields (or wrap around style) or goggles must be worn by all persons, including those already wearing prescription glasses.
Lab coat- knee length
Gloves appropriate to ALL chemicals you are using- not just the mercury compounds. Here are some choices of gloves that apply JUST to mercury compounds:
Elemental mercury — Silver Shield®, Responder®, Tychem® BR/LV, Tychem® SL, Tychem TK
Mercuric chloride — Tychem BR/LV, Tychem SL, Tychem TK
4. EMERGENCY PROCEDURES:
If you are splashed with a chemical in an area of your body which cannot be put under a sink faucet and flooded immediately — you must use an emergency shower. If your clothing is involved — remove it on the way to the shower:
Yell for help
Remain in the shower for 15 minutes
Get someone to call emergency for an ambulance
Do not re-don contaminated clothing.
If you are splashed in the eyes:
Yell for help
Hold your eyelids open with your fingers as you rinse your eyes with an eye-wash
Have your helper watch a clock for you to make sure that you continue to rinse your eyes for a full 15 minutes. All chemical exposures involving mercury compounds require immediate medical attention at hospital.
Mercury containing solutions must never be washed down the drain, and must be submitted to proper hazardous waste disposal facilities. Contaminated solid waste, including paper towel, pipet tips, syringes, and stock bottles must be disposed of as hazardous waste. Contaminated solid wastes should be collected in sealable disposable containers and stored in the fume hood until they can be removed from the darkroom.
IF YOU HAVE ANY DOUBTS, WHATSOEVER, ABOUT YOUR ABILITY TO SAFELY HANDLE TOXIC CHEMICALS, DO NOT USE MERCURIC CHLORIDE. But if you have a darkroom with facilities to safely store these materials and experience and proper protocols in handling them, mercuric salts offer one more tool in your darkroom toolbox. To once more quote Hill: “Let King Phillip’s motto be posted on all your employments — Remember thou art mortal”
TONERS AND INTENSIFIERS
As silver halide emulsions began to dominate the photographic industry at the turn of the 20th century, mercury would be more utilized as part of the chemical processing of images, primarily toners to intensifiers, which are their primary use in photography to the current day. At one stage, each of the major black and white film manufacturers, Kodak, Agfa & Ilford, published a formula for a mercury intensifier! They were known for their extreme action and minimal grain with modern thin film emulsions.
Although the exact mechanism of action is unknown, it has been suggested that in the presence of image silver, the mercuric(II) salt is reduced, liberating the halogen and allowing silver chloride or iodide to form and thickening the density, which may be complexed to form another compound in situ or redeveloped using a second developer, such as Kodak D-72 or sodium sulfite.
FORMULAS
Smith Victor VMI Mercury Intensifier
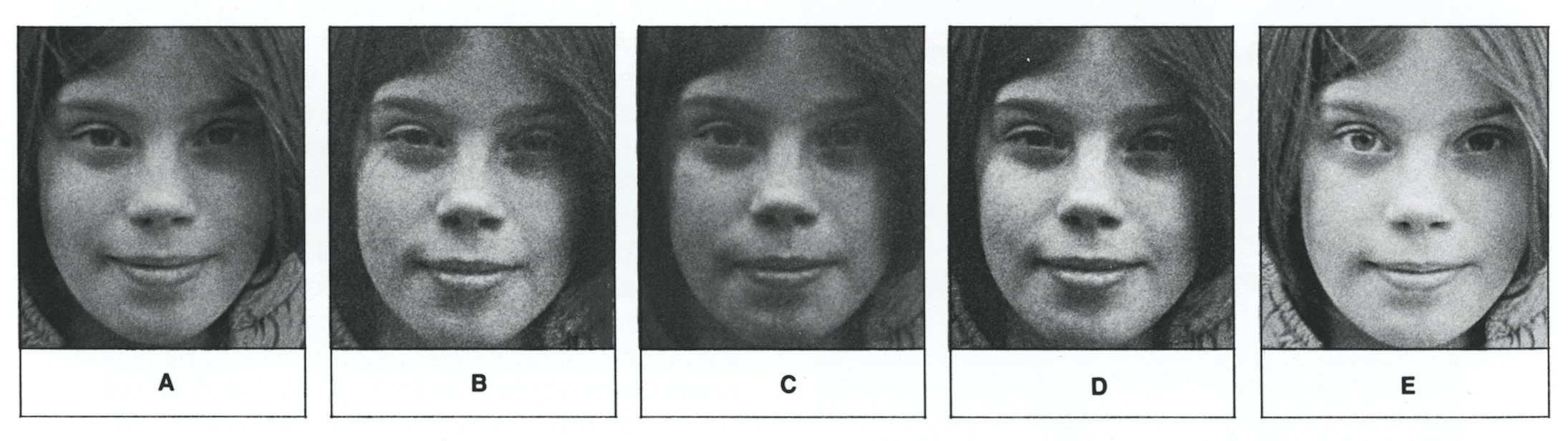
VMI is the most versatile of the extreme intensifiers. Chromium or silver intensifiers have limited intensifying capabilities. In my experience, their effect is inconsistent, often with little impact on modern, thin, fine grained emulsions, and difficult to control. Furthermore, because these intensifiers add metal to the existing silver grains, the grain pattern forms what I generally refer to as “bowling balls”. In contrast, VMI works beautifully and consistently on modern emulsions, particularly Kodak TMax films. It is a sub-proportional intensifier: its properties strengthen detail especially in the thin, weak areas areas of the negative, create greater separation of tone and of course, increase contrast, while avoiding adding density in the densest areas of the negative. It also creates beautiful grain without increasing the apparent size. Of course, as I photographer I always aim to make a perfect exposure, but occasionally I create a negative that’s underexposed; I have used this formula for this exact scenario with thin, under-exposed negatives that conventionally would be considered unprintable and irretrievable.
A comparison of prints from negatives intensified in common intensifiers and VMI, created by photographer George Krause. Half the roll was purposefully overexposed, and other half, underexposed. The film was underdeveloped in D-76, diluted 1:1 to produce a roll of flat negatives. All of the printys were developed in full-strength Dektol to reveal a more pronounced grain pattern.
(A) - A thin negative printed on Grade 6 paper. (B)- Made from a thin, flat negative that has been intensified with a copper intensifier for 10 minutes and printed on variable contrast paper, printed with a Grade 3 filter. (C) - Another thin, flat negative intensified with a chromium intensifier: bleached, cleared and redeveloped twice then printed on a variable contrast paper with a Grade 4 filter. (D) - A thin, flat negative intensified with VMI then printed on a variable contrast paper with a Grade 4 filter (E) - A dense flat negative intensified with VMI then printed on a variable contrast paper with a Grade 4 filter. Source: Lewis, Elanor (Editor): “DARKROOM”, p. 108, Lustrum Press, 1977
VMI can also be used to improve dense, flat negatives caused by overexposure, overdevelopment or both; the intensifier increases the contrast, relieving the flatness and improves the grain structure. But VMI’s use extends beyond fixing errors. Photographer George Krause used VMI extensively for negatives of normal density, to either change the grain pattern or to give more tonal separation: “Early in my career, I earned a living by printing the intaglio work of successful artists. I believe this had an effect on some of my earliest work, especially the dark prints I loved to make and still do. Black detail in a dark background is very exciting to me. Detail that would normally be lost in the dark areas is made visible with VMI. The VMI intensification process adapts the photographic grain pattern of either the dense or thin flat negative into a pattern that resembles aquatint or fine mezzotint.” Krause spoke personally to Mr. Smith after the Smith-Victor company had ceased manufacture, who shared the secret formula:
| Mercuric Chloride | 13g |
| Magnesium Sulfate | 60g |
| Potassium Iodide | 30g |
| Sodium Sulfite | 15g |
| Water to make | 1 litre |
I first encountered VMI while in University in 1994; I had just bought the first edition of Steve Anchell’s “Darkroom Cookbook”, and was immediately intrigued by its description of the intensifier. I was a second year scientific photography student and created underexposed negatives more than I care to admit. The sub-proportional action combined with the mezzotint grain was exactly what I was after. I asked the Faculty’s facilities manager, who was in charge of photographic chemistry, if they had mercuric chloride. I was surprised when he simply handed over a jar of 250 grams of mercuric chloride, saying they’d wanted to get rid of it for a while and were happy to see it go!
Returning to the darkroom, I set about preparing the chemistry. All components must be mixed in order. Once you add the potassium iodide, a bright orange precipitate will be formed: don’t be alarmed, this is normal! Continue to add the chemicals and mix, then let the solution stand for 15 minutes to allow the precipitate to settle. Finally, filter the solution and transfer to an amber bottle. Set aside the filtrate for proper hazardous waste disposal.
TO USE:
Prepare the negative. It should be hardened in a formalin hardener, such as KODAK Prehardener SH-1:
| Water | 500ml |
| Formaldehyde, about 37% solution | 10ml |
| Sodium Carbonate (Monohydrate) | 6g |
| Water to make | 1 litre |
An 8x10 negative intensified in VMI in 1996. This negative was originally 5 stops underexposed and unprintable. Once intensified, the image printed beautifully on conventional Grade 2 paper.
This formula is recommended for the treatment of negatives that normally would be softened by the chemical treatment given for the removal of stains or for intensification or reduction. Hardening may remove staining from pyro development, but so will the intensification process. After hardening for 3 minutes, negatives should be rinsed and immersed for 5 minutes in a fresh acid fixing bath. Wash the negative in water for 30min to one hour.
2. Immerse in the intensifier. The negative can be intensified for 5 seconds to 10 minutes, depending on the amount of intensification and the age of the VMI. When used fresh, the most practical intensification happens in the first 30 seconds. You will see little change while the negative is in the solution, so don’t wait to see the intensification. Take care not to over-intensify the negative, which can result in mottling. Every negative I have intensified in fresh VMI only needed 15-20 seconds to achieve a massive increase in density, so err on the side of caution. As the solution ages, intensification takes longer; I have a bottle that was mixed in 1992 that is still working, though it takes 5-10 minutes to fully intensify the negative.
3. Wash the negative. Transfer the negative back to the wash water. Over the next 10-15 minutes as the negative washes, it will turn bright orange. This is normal. Continue to wash for 30-mins to 1 hour. Photo-flo and dry as per usual. If the negative develops mottling of the image during washing, simply re-fix the image in a plain hypo bath, which will dissolve the intensification. Then simply wash the negative and start again.
4. Secure the solution. Once you’re done with the intensification, carefully decant the solution back into the storage bottle and secure in a lockable chemical cabinet. Once mixed, the solution lasts decades. Again, remember to never wash mercuric chloride solutions down the drain. They are long term environmental poisons and must be disposed of via professional hazardous material facilities.
The newly intensified negative is weakly light sensitive without further treatment. Therefore VMI intensified negatives must be permanently protected from the light when not being used, otherwise the colour mottles and discolours. An example may be seen in the image above, along the lower edge. This discolouration as a result of unintended but extended exposure to light on that area for a period of time. Alternatively, a final bath of 5% sodium sulfite will protect the image and make it permanent.
Modified Monckhoven’s: The Kodak IN-1 Mercury Intensifier
Charles Emanuel Désiré van Monckhoven (1834 – 1882) was a Belgian chemist, physicist, and photographic researcher, perhaps best remembered for the intensifier that bore his name. Monckhoven’s Intensifier is a bleach-redevelop style intensifier that permits extreme intensification with subproportional density. The original formula not only used mercury, but added potassium cyanide, which is a deadly poison by all routes of exposure; just 0.2g is sufficient to kill an adult in minutes! There are strict protocols and laboratory guidelines for using cyanides more complex than those listed above for mercury, as well as specific regulations for the purchase, possession, use and disposal of cyanide, which eliminates the practicality of the original formula for Monckhoven’s intensifier in the contemporary darkroom.
The formula of Kodak’s IN-1 is nearly identical to Monckhoven’s, except for Part B. Instead of bromide-cyanide-silver solution, you can use one of several choices of much less toxic redeveloping agents than cyanide, and reduces the number of deadly reagents to just mercuric chloride! The intensification is less intense, but is considerably safer to use and should still provide a significant contrast gain. Remember: the solution is still highly toxic, so the same guidelines for safety are required.
| PART A | Potassium Bromide | 22.5g |
|---|---|---|
| Mercuric Chloride | 23g | |
| Water to make | 1 litre |
| PART B choices | 10% solution sodium sulfite, or, |
|---|---|
| Kodak D-72, diluted 1:2, or, | |
| 10% ammonia |
Immerse the negative in Part A until completely bleached, wash, and then immerse in Part B, which will redevelop the image. The choices listed give progressively greater density in the order given. Where permanence of the resulting image is essential ammonia should not be used for redeveloping. Kodak documentation states that redevelopment cannot be controlled as with the KODAK Chromium lntensifier ln-4, but must go to completion. In contrast, other volumes state that the user can continue only until intended density is achieved. If you over-intensify, wash and treat in a weak alkaline hypo bath, which will reduce the density.
Mercury Toner for Iron-Silver processes
The addition of small amounts of mercuric chloride to the working sensitizer can modify the colour and tonal range of the final image and also, in combination with double toning, produce interesting split tones in the image. The formula is simple:
| Mercuric Chloride | 1g |
| Distilled Water | 30ml |
Add the working solution to the sensitizer at the ratio of about 1 part working solution to 20 parts sensitizer.
CONCLUSION
While the use of mercury presents undeniable challenges for the modern darkroom; from high toxicity and rigorous working precautions to stringent waste regulations, this unique element holds a special place in photochemistry. It is integral to image formation in modern daguerreotypy, and in the darkroom, its ability to salvage and enhance delicate or underdeveloped negatives makes it an invaluable tool in the hands of those who respect its hazards. When used with expertise and care, it can breathe new life into images that might otherwise be lost. While modern alternatives may offer safer or more convenient solutions, there remains a space for this historic intensifier—particularly in cases where no other method can achieve the same degree of recovery.
Mercury and its compounds are not for the casual darkroom hobbyist, but for those with the resources and capability to navigate its complexities, these materials stand as both a challenge and a rare opportunity. Whether you’re a contemporary daguerreotypist or a technician rescuing an irreplaceable negative, the possibilities of mercury are a testament to the enduring intersection of chemistry and photographic artistry.
REFERENCES
Wikipedia. (2024): “Mercury (element)” (2024) Available at: https://en.wikipedia.org/wiki/Mercury_(element) (Accessed: 19th June 2024).
Ware, Mike, “Cyanomicon II” (1999): Available at: https://www.mikeware.co.uk/downloads/Cyanomicon_II.pdf (Accessed: 2021).
Stulik , Dusan C. & Kaplan, Art : “The Analytical Signatures of Photographic Processes: Platinotype”. Available at: https://www.getty.edu/conservation/publications_resources/pdf_publications/pdf/atlas_platinotype_corrected.pdf , The Getty Conservation Institute, 2013
Nelson, Kenneth: “Mercury and the Daguerreotypist: A Modern Assessment”, Available at https://www.cdags.org/wp-content/uploads/Merc.Mod.Dagist.pdf, The Daguerreian Society Annual, 1994.
Barger, M. Susan and White, William B. : “The Daguerreotype: Nineteenth-Century Technology and Modern Science”, Washington London, Smithsonian Institution Press, 1991
Silvia A. Centeno, Franziska Schulte, Nora Kennedy, Alejandro Schrott: “The formation of chlorine-induced alterations in daguerreotype image particles: A high resolution SEM-EDS study” (DOI: 10.1007/s00339-011-6570-2), 2011
Pharmaceutical Journal Inset: “Photographic formulae”, p. 18, April 18, 1903
Anchell, S.: “The Darkroom Cookbook” , p. 246, Focal Press (First Edition), 1994.
Pénichon, Sylvie (2013): Twentieth Century Colour Photographs. The Complete Guide to Processes, Identification & Preservation. London, Los Angeles, Thames & Hudson, p. 235.
Coe, Brian (1978): “Colour Photography. The First Hundred Years 1840-1940”. London: Ash & Grant.
Kodak:“Processing Chemicals & Formulas for Black and White Photography” (Publication J-1, p. 42), Eastman Kodak Company, CAT 152 8462, 1977
Lewis, Elanor (Editor): “DARKROOM”, pp. 107-110, Lustrum Press, 1977
Towler, John. “The Silver Sunbeam”. Joseph H. Ladd, New York: 1864
Hill, Levi L.: “A Treatise on Heliochromy”, Available at: https://www.cdags.org/cdags_resources/A_Treatise_on_Heliochromy.pdf, Robinson & Caswell, New York, 1856
Ostroff, Eugene: “Pioneers of Photography: Their Achievements in Science and Technology”. The Society of Imaging Science and Technology, Northeastern University Press, Springfield, VA, 1984.
American Conference of Governmental Industrial Hygienists, “Threshold Limit Values for Chemical Substances and Physical Agents”, ACHIH, Cincinnati, Ohio, 2004
Bingham, E., Cohrssen, B., Powell, C., Patty: “Toxicology Fifth Edition”, Wiley Interscience, New York, 2001.
Board of Regents of the University System of Georgia, “Design Criteria for Laboratory Furniture and Fume Hoods”, 2000
Georgia Department of Natural Resources: “Public Employees Hazardous Chemical Protection and Right to Know Act of 1988”, Title 45 Chapter 22.
Georgia Institute of Technology Business and Finance Manual: “Environmental Health and Safety 8 Georgia Tech Lab Safety Manual”, Chapter 10.6, www.safety.gatech.edu
Klassen, Curdis D: “Casarett & Doull’s Toxicology: The Basic Science of Poisons”, Fifth Edition, McGraw-Hill, New York, 1996
Laurence Livermore National Laboratory: “ES & H Manual” Volume II Part 14.5 www.llnl.gov/es_and_h/hsm/doc_14.05/doc14-05.html
National Research Council: “Prudent Practices in the Laboratory”, Washington, D.C. 1995.
United States Environmental Protection Agency: Title 40, “Code of Federal Regulations”, Parts 260-273, Resource Conservation and Recovery Act
© Jesse Andrewartha 2024